|
Such a low standard atomic weight of aluminium has effects on the properties of the element. Īl is essentially the only isotope representing the element on Earth, which makes aluminium a mononuclidic element and practically equates its standard atomic weight to that of the isotope. No elements with odd atomic numbers have more than two stable isotopes even-numbered elements have multiple stable isotopes, with tin (element 50) having the highest number of stable isotopes of all elements, ten. 1995 - 2023.Main article: Radiation astronomy/Subatomics Periodic Table of Elements - Aluminum - Al. If you need to cite this page, you can copy this text: This database focuses on the most common chemical compounds used in the home and industry. Molar mass calculations are explained and there is a JavaScript calculator to aid calculations. Molar Mass Calculations and Javascript Calculator.Introduces stoichiometry and explains the differences between molarity, molality and normality. Related ResourcesĪnswers many questions regarding the structure of atoms. Common Chemical Compounds of Aluminum ReferencesĪ list of reference sources used to compile the data provided on our periodic table of elements can be found on the main periodic table page.So if you hear someone say "al-u-min'-i-um foil" instead of aluminum foil, you'll know where it came from. England and elsewhere in the world they still spell it aluminium. American Chemical Society changed the spelling back to aluminum in 1925, which we still use. Two years later it was changed to aluminium to conform with the "ium" in most other elements. In 1807 Davy proposed the name alumium for this undiscovered metal, but it wasn't until 1827 that Wohler actually isolated aluminum, though an impure form was isolated by Oersted two years earlier. Lavoisier thought that alum was an oxide of this undiscovered metal. de Morveau recognized the base in alum in1761 and proposed it be called alumine. Actually the ancient Greeks and Romans used alum (aluminum sulfate with potassium) in medicine and in dying. 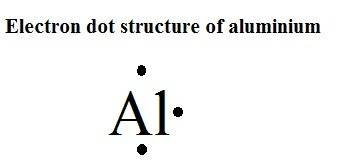
Additional Notes: While aluminum was discovered by Hans Christian Oersted, Denmark, 1825 (impure form) most credit Wohler with isolating it in1827.Alloys containing copper, magnesium, silicon, manganese and other metals are much stronger and more durable than aluminum, making aluminum useful in the manufacture of aircraft and rockets. Uses of Aluminum: Kitchen utensils, building decorations, electrical transmission (not nearly as conductive as copper, but cheaper).Main mining areas are Surinam, Jamaica, Ghana, Indonesia and Russia. Total world production is around 15,000,000 tons per year. Obtained by electrolysis from bauxite (Al 2O2). Sources of Aluminum: Most plentiful metal in earth's crust (8%), but never occurs in free form.70kg human: 60 mg Who / Where / When / How Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances. Target Organs: Eyes, skin, respiratory system.Routes of Exposure: Inhalation Skin and/or eye contact.TWA: 10 mg/m 3 total particulate 5 mg/m 3 respirable particulate.TWA: 15 mg/m 3 total particulate 5 mg/m 3 respirable particulate.Vapor Pressure = C Regulatory / Health.Flammablity Class: Combustible Solid, fine dust is easily ignited.Enthalpy of Vaporization: 293.7 kJ/mole.Enthalpy of Atomization: 322.2 kJ/mole 25☌.Description: Silvery, light weight, non-magnetic, non-sparking, malleable metal.Conductivity Electrical: 0.377 10 6/cm Ω.Coefficient of lineal thermal expansion/K -1: 23.03E -6.Valence Electron Potential (-eV): 80.7 Physical Properties of Aluminum.Incompatibilities: strong oxidizers & acids, halogenated hydrocarbons.Electronegativity: 1.61 (Pauling) 1.47 (Allrod Rochow).Electrochemical Equivalent: 0.33556g/amp-hr.Valence Electrons: 3s 2p 1 Electron Dot Model.Number of Neutrons (most common/stable nuclide): 14.Number of Electrons (with no charge): 13.Electrons per Energy Level: 2,8,3 Shell Model.Electron Configuration: 1s 2 2s 2p 6 3s 2p 1.Crystal Structure: Cubic face centered.Cross Section (Thermal Neutron Capture)σ a/ barns : 0.232.Swedish: Aluminium Atomic Structure of Aluminum.Series: Metals Aluminum's Name in Other Languages.Common Chemical Compounds of Aluminum Overview of Aluminum.In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies. Common chemical compounds are also provided for many elements. 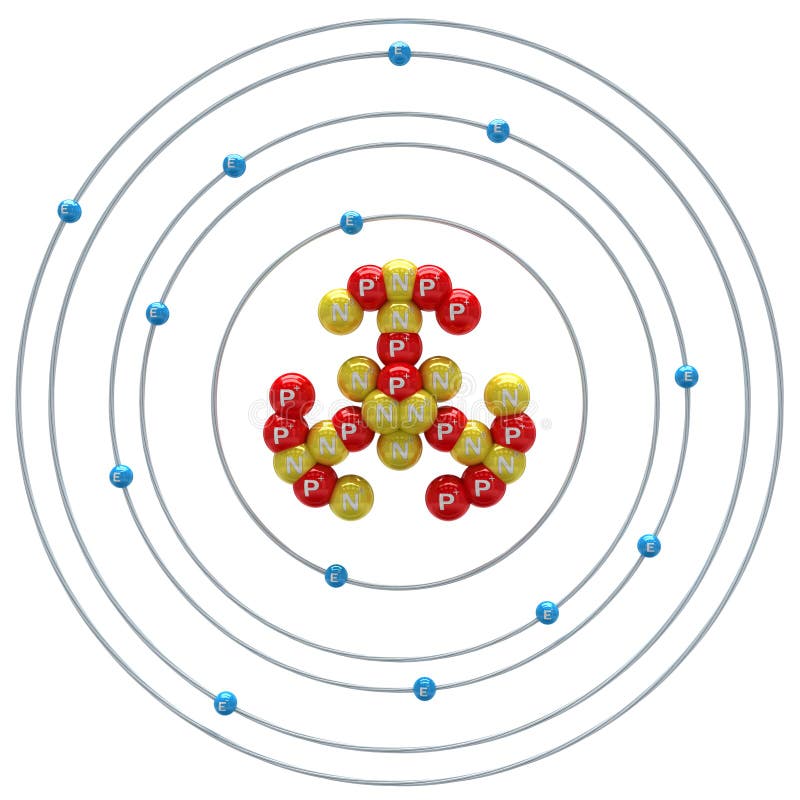
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
